This study introduces a novel computational approach that allows the inference of DNA methylation patterns in ancient, non-skeletal tissues, particularly the brain, using only bone-derived ancient DNA. DNA methylation is a critical epigenetic mechanism influencing gene regulation and cell identity, playing a crucial role in evolution. Traditionally, studies of ancient DNA have been restricted to skeletal tissues, leaving direct observation of the brain’s epigenetic patterns largely inaccessible due to its absence from the fossil record. Hence, while human ancient genomics is well-studied, the evolution of epigenetic marks that may have influenced human brain evolution are little known.
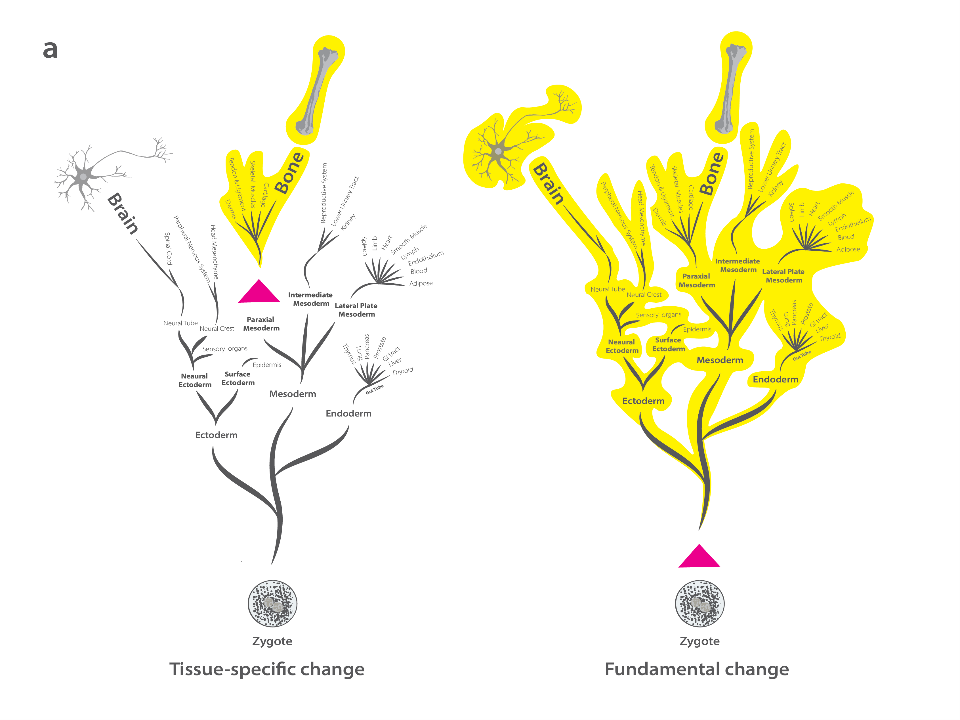
By leveraging the fact that some DNA methylation patterns are established early in embryonic development and maintained across multiple tissues, the authors developed an algorithm that predicts “fundamental” methylation changes—those likely shared between bone and brain. Using a validation dataset from modern primates, the method was shown to have high precision in predicting methylation states across tissues and was subsequently applied to archaic humans (Neanderthals and Denisovans).
The key finding is the identification of over 1,800 genomic positions in prefrontal cortex neurons of archaic humans that exhibit methylation differences compared to modern humans or chimpanzees. These positions are linked to hundreds of genes, many of which have known neural functions. Notably, many changes were found within the neuroblastoma breakpoint family (NBPF) genes that are associated with brain volume expansion and cognitive traits in humans. This sheds new light on epigenetic regulation and the role it may have played in human brain evolution.
This work opens a new avenue for paleoepigenetics, allowing neuroscientists to explore regulatory changes in ancient brains, with potential implications for understanding the molecular basis of human-specific cognitive traits and neurodevelopmental disorders.
B.
A: Conceptual framework of the algorithm.
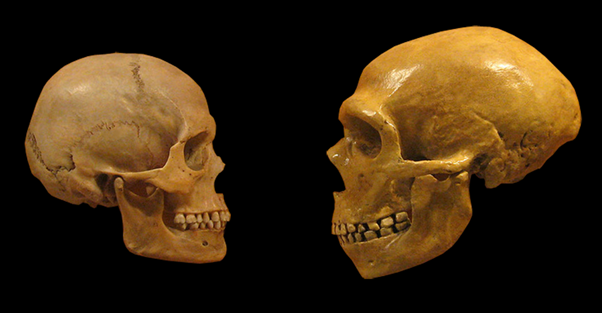
B: Comparison of Modern Human and Neanderthal skulls from the Cleveland Museum of Natural History.

